 Parkinson’s disease affects around one million people in the US and between seven and ten million worldwide.
Parkinson’s disease affects around one million people in the US and between seven and ten million worldwide.
In people with Parkinson’s disease, the neurons in the brain that produce dopamine die off. Dopamine is a neurotransmitter, a chemical that helps send messages in the brain. It is involved in many functions such as movement, reward, memory, sleep regulation, nausea, motivation and arousal and many more. Thus a reduction in dopamine can affect a person in many ways.
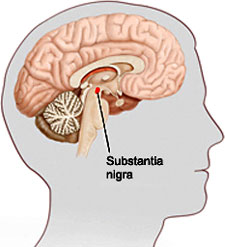
Substantia Nigra
People with Parkinson’s disease lose cells in the substantia nigra. The substantia nigra is part of the mid brain and is the area of the brain that produces a large amount of dopamine. It is also an area involved in movement. But the dopamine that this 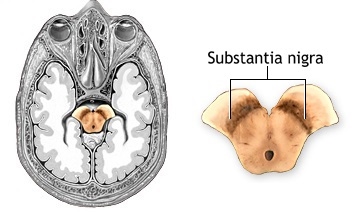 area produces moves throughout the brain and is used by areas involved in learning and emotion.
area produces moves throughout the brain and is used by areas involved in learning and emotion.
One treatment for Parkinson’s disease is medication that contains dopamine, to replace or enhance the low or missing levels of dopamine in the brain.
But researchers have been working to understand the processes by which neurons in the brains of people with Parkinson’s disease die.
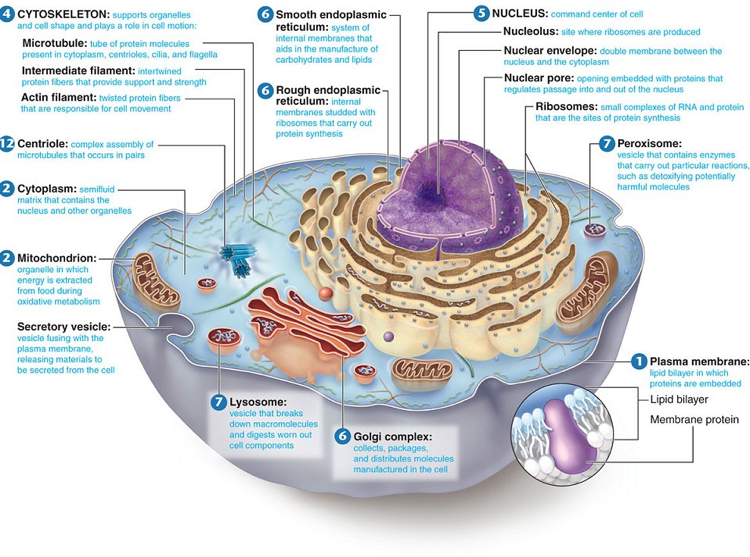
To understand some of this research, it is important to remember some of the structures of cells throughout the body and how cells create proteins.
We introduced cells in a previous post called Understanding Cells.
Cells are composed of a nucleus and smaller structures called organelles. The nucleus and three types of organelles are important in the creation of proteins.
Nucleus
The nucleus contains the blueprints for protein creation in the genetic code.
There are rare forms of Parkinson’s disease in which just one faulty copy of a gene can cause it. The Alpha-synuclein (SNCA) and the LRRK2 are examples. Two faulty genes (autosomal recessive)- PARK7, PINK1 or PRKN– can cause Parkinson’s disease. There are also genes, like altered GBA, that increase the risk of Parkinson’s in some families.
Organelles
Creation of proteins occurs in an organelle called the ribosome. If there is a faulty genetic code or if there is a mistake in the creation of the protein in the ribosome—like a problem in the way it is folded– there is a 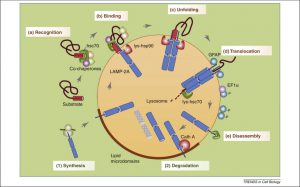 process in place to get rid of the damaged protein. Molecules in the cell, called chaperones, watch out for damaged proteins. If a chaperone finds one, it moves the damaged protein to the lysosomes. There, it is broken down and any useful parts are recycled.
process in place to get rid of the damaged protein. Molecules in the cell, called chaperones, watch out for damaged proteins. If a chaperone finds one, it moves the damaged protein to the lysosomes. There, it is broken down and any useful parts are recycled.
Research by Cuervo and her associates have clarified problems with this particular process called CMA (for chaperone-mediated autophagy). Sometimes this process is blocked. In Parkinson’s disease, when the process is stopped or reduced, proteins are not being broken down. They accumulate in clumps.
Mitochondria have the primary role of energy productions. They are also involved in the use of calcium and also in generating oxides or free radicals abut also in utilizing anti-oxidants to detoxify them. The mitochondria maintain a balance with in the cells.
Research described in Science in 2017 by Burbulla and her associates explains some of what happens over  time in the neurons leading to Parkinson’s disease. Their research has tracked the development of disease producing biochemical changes that start with the mitochondria. An imbalance occurs with the number of free radicals and anti-oxidants. Oxidized dopamine accumulates in the neurons leading to a reduction in the production of enzymes (glucocerebrosidase) that break down lipids (glucocerbrosides). This leads to problems in lysosomes (like described above) and the accumulation of alpha synuclein (a protein found in healthy neurons). Most people with Parkinson’s disease have Lewy Bodies, or clumps of alpha synuclein.
time in the neurons leading to Parkinson’s disease. Their research has tracked the development of disease producing biochemical changes that start with the mitochondria. An imbalance occurs with the number of free radicals and anti-oxidants. Oxidized dopamine accumulates in the neurons leading to a reduction in the production of enzymes (glucocerebrosidase) that break down lipids (glucocerbrosides). This leads to problems in lysosomes (like described above) and the accumulation of alpha synuclein (a protein found in healthy neurons). Most people with Parkinson’s disease have Lewy Bodies, or clumps of alpha synuclein.
Both of these research areas are clearing up what is happening on a cellular level in Parkinson’s disease. One of the areas of active research is in developing therapies that will restart, intervene or increase the speed of protein recycling, another area is in creating interventions to stop mitochondrial oxidative stress.
We will address other areas of research and upcoming treatment therapies in a future post.






Thank you for this information. I think understanding the cause is key to developing treatment. This article gives me hope.
What’s your take on ultra accel II for mitochondrial health?
Very nice explaination. I am trying to collect maximum information on PD and role of Naturopathy and Acupuncture.pl give me more information on PD on my email id.Thanks a lot
Great article understanding dopamine function and PD.
would like to know aboup PSP (progressive supranuclear palsy) and domine or any other research as this is also mid brain problem
I did a lot of exercises as I was advice by a specialist. I had Sinemet for some years but nothing happened but my stooped posture, tremors and other parkinsons diseases symptoms remains till i was introduce to madidaherbaclinic.weebly.com were I got the PD herbal formula which I used early last year and this is another year again marking a year free of Parkinson disease. Madida herbal formula totally reverse my PD. Your can visit their website for more update about them
Im actually suffering from my usual insomnia issues,and I’ve been doing a lot of research tonight! I actually suffer from bipolar disorder, as well as chronic anxiety, as well as ptsd. I thought my rbd symptoms for about a decade or even longer, in which I scream out, as well as punch and kick, were directly related to my traumatic past which led to my ptsd- I’ve in actuality, broken a toe, bruised my shins; an unbelievable amount of times, as well of caused many disturbances to my poor wife who has to deal with all the this for most of our 24 year to the date marriage, and bless her soul for being a such a compassionate person, but I’m truly beginning to think that I may in actuality be showing early signs of Parkinson’s disease. I’m about to turn 44, and in the last year have developed, a twitching, or jerking motion in my thigh, hip, shoulder,and neck, sometimes throughout the day, but more so when I lay down at night. I’ve noticed a pill rolling term that they mentioned in this video, where in the last 6 months I’ve developed an unusual habit of moving my fingers against my thumb uncontrollably. Does anybody have relation to these issues,and what steps should I be taking, because I’m a little concerned that in 15 years from now I may be suffering from full blown Parkinson’s; a little worried, just need some advice…
blessingway121