Oncology Basics 2016 continues with a look at the immune system and immunotherapy. As described in a previous post, one of the regulators of cell division and proliferation is the immune system. Harnessing the power of the immune system to attack and destroy cancer cells is a promising area of oncology research.
Immune System Cells
Below is a graphic to help understand the basic components of the immune system. The first cell (the green stem cell) is in the bone marrow. It can become either a lymphoid stem cell (blue) or a myeloid progenitor (yellow).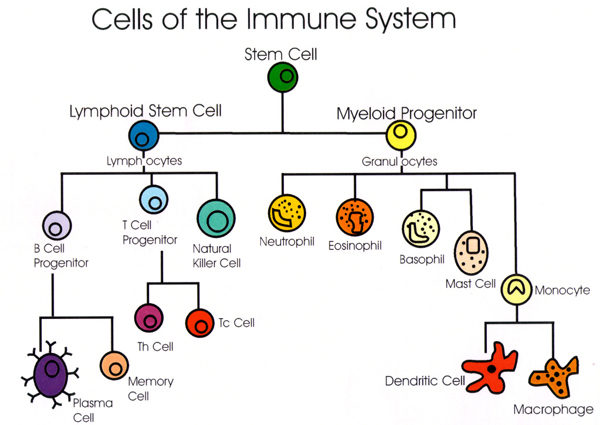
Innate Immune System
The first line of the first line of defense against bacteria and other microorganisms is the innate immune system. The myeloid progenitors create the innate immune system, made up of a group of cells called granulocytes. Once a foreign substance enters the body, cells of the innate immune system work to remove that substance, recruit other cells to the area by releasing cytokines, and activate the adaptive immune system.
Adaptive Immune System
The adaptive immune system spring from the lymphoid stem cells. Lymphocytes evolve either in the bone marrow–called B-cells–or in the thymus–called T-cells.
The innate immune system is important in instigating and directing the adaptive immune cells responses. Dendritic cells and macrophages are the links between the innate and adaptive immune system.
responses. Dendritic cells and macrophages are the links between the innate and adaptive immune system.
Immature dendritic cells are found in all tissues and their purpose is to sample their environment for foreign substances or antigens. When they connect with those substances, they quickly mature becoming Antigen Presenting Cells (APCs). They then go to the lymph nodes where they activate T-cells and B-cells.
T-cells become T-helper (Th) cells and cytotoxic (Tc) T-cells. Cytotoxic means that it kills cells that are damaged. When B-cells are presented with an antigen, they change into a plasma cells and creates antibodies to the antigen.
Much research has been done on activating the adaptive immune system to seek out cancer cells. Here are a few of the strategies being used.
Monoclonal Antibodies
Antibodies (like those produced by B-cells) can be designed in the lab to target antigens that are found on cancer cells. These are called monoclonal antibodies. Trastuzumab (Herceptin®) is an example of a monoclonal antibody used to treat breast cancer. Below is a video by the Cancer Research Institute describing monoclonal antibodies.
Check-point Inhibitors
Antibodies are also used to intervene in certain pathways that have been hijacked by cancer cells. The cancer cells hide from the immune systems normal functions of destroying over-proliferating cells. One of the pathways they hijack is the programmed death (PD) pathway or apoptosis mentioned as one of the natural controls of over proliferation in a previous post.
One of the jobs of the T-cells is to check proteins on the surfaces of cells to determine if the cell is normal. If the cell is infected or cancerous, the T-cell destroys it. To stop T-cells from destroying them, cancer cells produce a protein called PDL-1, an immune checkpoint molecule that stops T-cells from destroying it. Researchers have fashioned antibodies, called checkpoint inhibitors, to intervene in this process. Ipilimumab (Yervoy), Nivolumab (Opdivo) and Pembrolizumab (Keytruda) are examples of these drugs.
Below is a video by Dana Farber Cancer Institute to describe checkpoint inhibitors.
Adoptive Cell Therapy
Researchers have found that there are lymphocytes in tumors that are trying to fight the cancer called tumor-infiltrating lymphocytes or TILs. In this immunotherapy strategy, these TILs are harvested from the person’s tumors and tested to see which are the most effective in killing the cancer cells. Those are grown in the lab and then put back into the patient.
To prepare the patient for the infusion of so many TILs, the patient has to go through high dose chemotherapy for lymphodepletion.
CAR T-cell Therapy
A twist on adoptive cell therapy is called chimeric antigen receptor T-cell therapy. The T-cells are removed from the patient and are genetically re-engineered to produce chimeric antigen receptors on their surfaces. These protein receptors help T-cells recognize antigens on cancer cells.
After the re-engineering, the CAR T-cells are grown in the lab and once millions have grown the patient receives them in an infusion. They grow in the body and attack the cancer cells. These cells remain circulating in the body and help to guard against cancer recurrence.
Here is a video by the Cancer Research Institute describing ACT.
Oncolytic Viruses
This strategy involves genetically modifying viruses so that they kill cancer cells. The virus is injected into the cancer; it does not enter healthy cells. It starts to make copies of itself and causes the cancer cells to explode. Antigens are released into the surrounding tissue which are taken up by immature dendritic cells and activate T-cells and B-cells to target cancer cells. T-VEC (talimogene laherparepvec–Imlygic) is the name of the first oncolytic treatment. Here is a video from Cold Genesys.
Promising News From ASCO 2016
One study being presented at American Society of Clinical Oncologists (ASCO) Meeting 2016 describes the use of adoptive cell therapy in people with metastatic melanoma. In a clinical trial 24% of those treated with ACT had a durable complete regression and median survival of more than 3 years. The amount of preparatory lymphodepletion– chemotherapy alone or with total body radiation–didn’t change this result.
In another study, patients with relapsed or difficult to treat blood cancers (acute lymphocytic leukemia, chronic lymphocytic leukemia and non-hodgkins lymphoma) were treated with CAR T-cell therapy. Again different types of preparatory lymphodepletion were being tested. Those who received two forms of chemotherapy did better but importantly a “durable” complete remission was achieved in a “high fraction” (quote from abstract) of those treated.
Hope
 The first post of this series began with a discussion between President Nixon and AMA president. At the meeting, Hoffman recalled saying, “I think every physician who is dedicated and sincere never goes on the floor of a hospital where he has cancer patients…-without feeling helpless…I think of the millions of hours and the millions of dollars spent on cancer research and how little progress we have made.”
The first post of this series began with a discussion between President Nixon and AMA president. At the meeting, Hoffman recalled saying, “I think every physician who is dedicated and sincere never goes on the floor of a hospital where he has cancer patients…-without feeling helpless…I think of the millions of hours and the millions of dollars spent on cancer research and how little progress we have made.”
It is safe to say that, in the 45 years since this interaction, enormous progress has been made in understanding cancer: how it starts and what happens in cancer cells and the immune system.
In the abstracts presented at ASCO 2016, the huge advances in molecular biology, microbiology and computer capacity are making a difference. Who could have imagined in 1971 that it would be possible for scientists to engineer molecules to attach to T-cells that specifically target cancer cells? And that, with this one treatment strategy, people who had metastatic cancer are now surviving with “durable complete regression” of their cancer.
There is room and reason for hope.






I have autoimmune disease (Dermatomayositist) I am on Methotrexate, Mycophynalate, Perdnislone,
due to my medication I had put too much wait. Advise how to control my weight.
Hello, We appreciate your comment. However, since Medivizor does not practice medicine nor provide Medical advice, we cannot directly respond to your comment. Please discuss this concern with your physician.